
Advancing the development of novel antidepressants and anxiolytics based on clinically validated mechanisms using our Glyph™ platform
Powered by our Glyph platform we are advancing first and best-in-class medicines for people living with neuropsychiatric disorders.
Advancing the development of novel antidepressants and anxiolytics based on clinically validated mechanisms using our Glyph™ platform.
PHASE 1
PHASE 2
PHASE 3
PRE-CL
PH 1
PH 2
PH 3
GlyphAllo™ (SPT-300)
Glyph Allopregnanolone
Major Depressive Disorder (MDD)
Designed to overcome oral bioavailability limitations
GlyphAgo™ (SPT-320)
Glyph Agomelatine
Generalized Anxiety Disorder (GAD)
Designed to reduce or eliminate need for liver function testing
Glyph2BLSD™ (SPT-348)
Glyph 2-bromo-LSD
Multiple Indications*
Designed to improve PK & tolerability
1 The FDA and corresponding regulatory authorities will ultimately review our clinical results and determine whether our investigational product candidates are safe and effective. No regulatory agency has made any such determination that our investigational therapies are safe or effective for use by the general public for any indication.
* Depressive disorders, including treatment-resistant depression (TRD), post-traumatic stress disorder (PTSD), and headache disorders with significant unmet need.
Glyph aims to unlock the therapeutic potential of drugs in CNS and beyond.
In addition to our three lead candidates – we have robust discovery programs and multiple pipeline programs underway

We aim to develop novel, leading treatment options that will make a significant impact for patients and their families


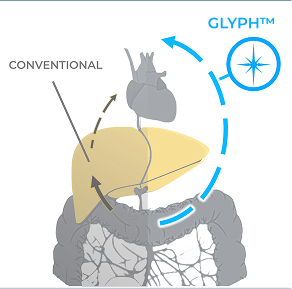
Our Glyph™ Platform Leverages the Lymphatic System to Unlock New Medicines Based on Clinically Validated Mechanisms
Proprietary platform advances active drugs previously limited by high first-pass metabolism, low bioavailability, and/or side effects

Glyph™ is based on the pioneering research of the Porter Research Group at Monash University in Melbourne

Christopher Porter, Ph.D.
Original Co-inventor of Glyph Technology, Director of the Monash Institute of Pharmaceutical Sciences
The group and it’s collaborators have published research in multiple publications supporting the Glyph™ platform’s capabilities.
Seaport holds a worldwide exclusive license with a right to sublicense under Monash University’s intellectual property rights related to the Glyph platform to develop and commercialize all of our product candidates and discovery programs.
Key co-inventors of the breakthrough technology include scientists who are now members of the Seaport team

Jamie Simpson, Ph.D.
Original Co-inventor of Glyph Technology, Head of Chemistry at Seaport
Dan Bonner, Ph.D.
Co-founder, Senior Vice President, Platform at Seaport

Members of our team co-founded and were central to the success of Karuna Therapeutics, which invented and advanced the first-in-class medicine Cobenfy (formerly known as KarXT), and was acquired by Bristol Myers Squibb in March 2024 for $14 billion.
Cobenfy was approved by the FDA in September 2024 and is the first drug to provide a new mechanism of action for schizophrenia in decades.
The approach we’re taking at Seaport has some important similarities to what the team did at Karuna. We are identifying drugs with proven efficacy, addressing the limitations that have held those drugs back, and then designing and executing studies with an experienced team. At Seaport, we also have distinct advantages that Karuna didn’t have, including our Glyph platform, a robust pipeline, and novel composition of matter IP.
Validated Efficacy
Xanomeline had generated exciting efficacy data at Eli Lilly, under the leadership of Dr. Steve Paul, but the drug was not being advanced due to GI tolerability issues.
These challenges were later addressed by members of our team through the invention of KarXT (now called Cobenfy) at PureTech – which was led by Daphne Zohar. Karuna was co-founded by Ms. Zohar who brought in Dr. Paul as CEO.

Proven Team
Cobenfy (formerly KarXT), was developed by combining xanomeline (a muscarinic receptor agonist) with trospium (peripherally acting antagonist that does not cross the blood-brain barrier).
Karuna ran 3 successful registration-enabling studies, achieved FDA approval, and was a success for shareholders with Karuna’s $14 billion acquisition by Bristol Myers Squibb.
Most importantly, Cobenfy represents the first new mechanism for people with schizophrenia in decades.
Karuna's KarXT Case Study
Members of our team co-founded and were central to the success of Karuna Therapeutics, which invented and advanced the first-in-class medicine Cobenfy (formerly known as KarXT), and was acquired by Bristol Myers Squibb in March 2024 for $14 billion.
Cobenfy was approved by the FDA in September 2024 and is the first drug to provide a new mechanism of action for schizophrenia in 70 years.
The approach we’re taking at Seaport has some important similarities to what the team did at Karuna. We are identifying drugs with proven efficacy, addressing the limitations that have held those drugs back, and then designing and executing studies with an experienced team. At Seaport, we also have distinct advantages that Karuna didn’t have, including our Glyph platform, a robust pipeline, and novel composition of matter IP.
Validated Efficacy
Xanomeline had generated exciting efficacy data at Eli Lilly, under the leadership of Dr. Steve Paul, but the drug was not being advanced due to GI tolerability issues.
These challenges were later addressed by members of our team through the invention of KarXT (now called Cobenfy) at PureTech – which was led by Daphne Zohar. Karuna was co-founded by Ms. Zohar, and Dr. Paul later joined as CEO.
Proven Team

